Chronic limb ischaemia
Notes
Overview
Chronic limb ischaemia (CLI) refers to the development of inadequate perfusion to the lower limbs.
CLI is characterised by chronic narrowing of peripheral arteries resulting in symptoms of intermittent claudication, rest pain and may threaten limb viability. It can affect the upper limbs, but lower limb disease is far more common. CLI of the lower limb will form the focus of this note.
Pathogenesis
Chronic limb ischaemia is most commonly the result of atherosclerosis in the arteries supplying the lower limbs.
Atherosclerosis is an inflammatory process that causes narrowing of blood vessels. It involves endothelial injury, local inflammatory response and accumulation of low-density lipoproteins (LDL) resulting in plaque formation within arteries. Atherosclerosis of the coronary vasculature leads to acute coronary syndrome, of the peripheral vessels leads to peripheral vascular disease and of the cranial vasculature leads to ischaemic strokes.
The common risk factors for atherosclerosis means patients with peripheral vascular disease are at increased risk of other cardiovascular diseases (including stroke and MI).
Risk factors
Smoking and diabetes are the two most significant risk factors for developing peripheral vascular disease.
The risk factors predominantly relate to the development of atherosclerosis.
- Smoking
- Diabetes
- Age
- Family history of vascular disease
- Hypertension
- Hyperlipidaemia
- Obesity
- Raised homocysteine
It appears patients of black ethnicity have a greater risk of chronic limb ischaemia, though this may be in part due to the higher incidence of factors described above. Of note, in western countries, those of black ethnicity are at greater risk of complications and amputation (see here for more details). Further research is needed to understand and combat these racial disparities.
Clinical features
Early LEAD is typically asymptomatic, with intermittent claudication commonly the first symptom experienced.
Intermittent claudication
Cramp-like muscular pain in the lower limbs (typically affecting the calves). Symptoms are triggered by exercise / walking and relieved by rest.
Foot pulses may be absent and there is increased risk of non-healing skin injuries. Hair loss in the lower limbs may be evident. Erectile dysfunction is common.
Critical / limb threatening ischaemia
Rest pain, pain experienced at rest, heralds the development of critical / limb threatening ischaemia. Pain is often worse at night due to reduced action of gravitational forces, characteristically the patient hangs a leg of the edge of the bed to relieve symptoms.
Foot pulses are often absent. Ulcers, infections, gangrene and tissue loss may all occur.
Investigations
An ankle-brachial index is the first-line investigation in those with suspected lower extremity artery disease.
Ankle-brachial index
An ankle-brachial index (ABI) is typically the first-line investigation. It compares the blood pressure at the ankle and the arm. The ABI is calculated by dividing the ankle systolic BP by the arm systolic BP.
A low ABI (< 0.9) is indicative of lower extremity artery disease. It is not a perfect test, those with borderline results, or in whom suspicion persists further tests are required.
High ABI (> 1.40) is indicative of vessel wall calcification. Alternative tests like doppler US and toe brachial index (TBI) may be used.

Treadmill test
Treadmill is used to identify those with peripheral artery disease. Strandness protocol involves walking at 3 km/h on a 10% slope.
The test can be continued until the patient needs to stop due to pain. A decrease in ankle systolic BP of > 30 mmHg or ABI decrease > 20% is diagnostic of lower extremity artery disease.
Imaging
Duplex US: May be combined with ABI and exercise tests to diagnose lower extremity artery disease.
CT angiography: Offers good views of vessels, as well as identifying aneurysms and previous interventions. Involves radiation and contrast (consider renal function and sensitivity).
MR angiography: Offers good views of vessels and may be useful in identifying stenosis in calcified vessels. No radiation, but costly and time consuming. Metal stents / prosthesis may distort images.
Interventional
Digital subtraction angiography: An invasive technique that utilises catheter guided contrast injection combined with fluoroscopy and digital subtraction to isolate the vessels.
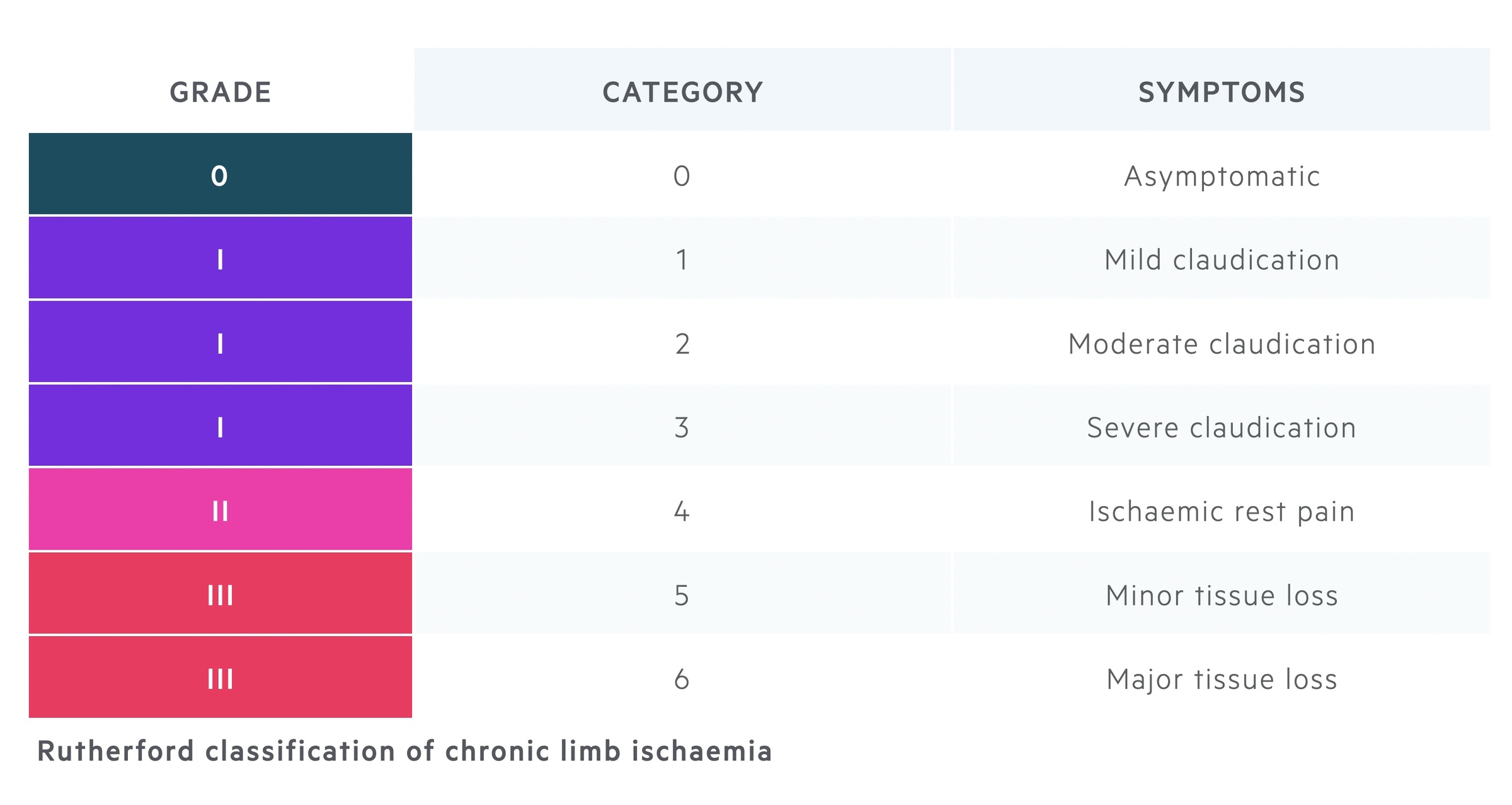
Rutherford classification
The Rutherford classification can be used to grade chronic limb ischaemia.
Conservative measures
Conservative measures are aimed at improving symptoms, reducing disease progression and addressing cardiovascular risk factors.
The secondary prevention of cardiovascular disease is important to improve the long-term outcomes in patients with peripheral vascular disease. This involves addressing modifiable risk factors and commencing pharmacological therapies. All patients should be screened for related conditions including hypertension and diabetes and treated accordingly.
- Address modifiable risk factors: smoking cessation is of the utmost importance. Dietary advice and appropriate weight loss should also be supported.
- Supervised exercise programme: these plans should be offered to patients with intermittent claudication. Plans should take into account an individual's abilities and co-morbidities. Excercise typically involves walking until the onset of symptoms, and where possible until the maximal pain is reached.
- Pharmacological therapies:
- Lipid therapy: statins should typically be offered to patients with peripheral vascular disease.
- Anti-platelet therapy: all patients with peripheral arterial disease should be offered Aspirin or Clopidogrel as secondary prevention.
- Naftidrofuryl oxalate: this is a peripheral vasodilator that may be considered in patients in whom conservative measures have failed and decline surgical options.
Revascularisation
Revascularisation techniques can be categorised as surgical or endovascular.
Revascularisation should be considered in patients with intermittent claudication who have failed conservative measures. All patients with critical limb ischaemia (where appropriate) should be urgently referred for review and discussion at the vascular MDT.
Choice of intervention is complex and nuanced. It is based upon the pattern of disease and patient based factors. Here we outline some of the options that may be considered.
Endovascular
Angioplasty +/- stent: Balloon angioplasty involves the opening up of areas of stenosis often followed by stent deployment.
Remote endarterectomy: Considered a hybrid procedure, involves the removal of plaque from within a vessel, often combined with stent.
Surgical
Bypass: Vessel (synthetic or autologous) is used to bypass the diseased segment of artery.
Prognosis
The majority of those with intermittent claudication will develop stable disease.
Diabetes is associated with a significantly increased risk of amputation by at least 50%. In patients with chronic limb ischaemia, symptoms can be progressive in up to a third, but generally the rate of major amputation is 1-3%. Patients who develop critical limb ischaemia (i.e. pain at rest) are at an increased risk of complications and amputation.
Cardiovascular disease is rarely isolated to the peripheries and so risk of stroke and MI are increased. Patients with peripheral arterial disease are 3 times as likely to die from cardiovascular diseases.
Last updated: October 2024
Have comments about these notes? Leave us feedback
